Biorecycling context and temperature constraints
Enzymatic recycling approaches target polymer bonds in PET, commonly used in packaging and textiles. Efficient depolymerization occurs near 70 °C, where the polymer becomes more accessible. Under these conditions, enzymes must retain global structural integrity while preserving flexibility at the active site. This combination is typically difficult to achieve.
Thermophilic enzyme model and analytical approach
The research team analyzed a cutinase derived from the fungus Chaetomium thermophilum. Both the native enzyme (CtCutWT) and a mutant variant (CtCutS136A) were examined. Structural characterization and thermal stability measurements were conducted using differential scanning calorimetry across a temperature range from 30 °C to 100 °C.
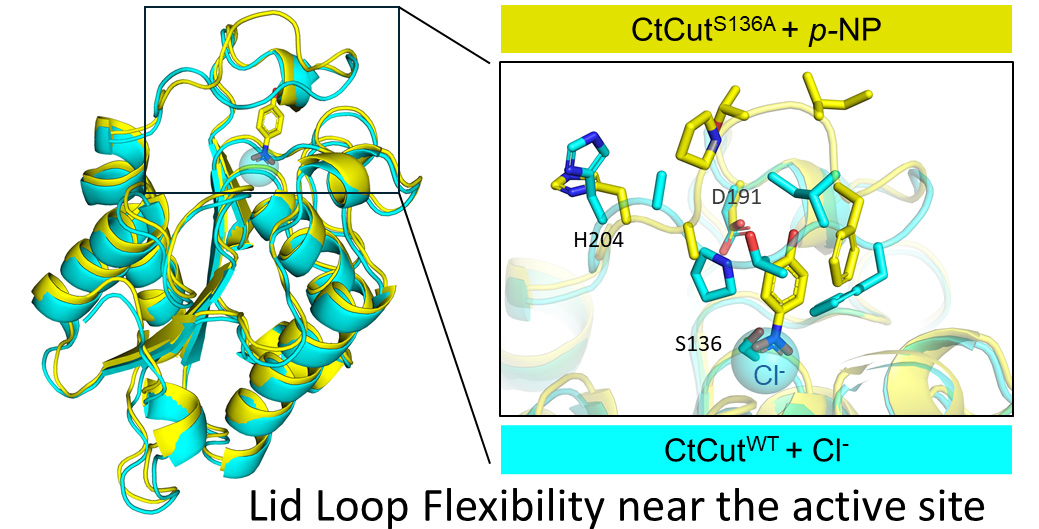
Rigid core and flexible lid loop
The enzyme adopts an α/β-hydrolase fold, providing a stable structural framework. A mobile lid loop covers the active site and regulates substrate access. Structural observations indicate that this loop undergoes conformational changes during ligand binding, enabling catalytic function without compromising overall stability.
A chloride ion detected near the active site suggests the presence of a positively charged microenvironment. This may support substrate interaction even in the absence of bound molecules.
Stepwise thermal unfolding
Thermal analysis identified a two-stage unfolding process, with initial structural changes beginning at approximately 60 °C and a second transition occurring between 65 °C and 70 °C. These findings indicate that different regions of the enzyme exhibit distinct thermal stabilities, supporting the concept of functional segmentation within the protein.
Implications for enzyme engineering
The results demonstrate that effective enzymes for PET biorecycling may require a stable core structure combined with localized flexibility at the catalytic interface. This structural arrangement supports both heat resistance and substrate adaptability, offering a basis for the design of improved enzymes for industrial plastic recycling applications.
The study was published in the journal Crystals (Volume 16, Special Issue 4) on March 24, 2026.